
It is also suggested that the orientations of the methoxy-groups are influenced by the requirement of good matching between the p orbitals on oxygen and d orbitals on phosphorus. Oxygen is a diatomic molecule with linear molecular geometry and bond angles of 180 degrees.

The relative orientations of the 3 d orbitals on the phosphorus and 2 p orbitals on the nitrogen and oxygen atoms, as deduced from the molecular geometry, are used to set up π systems in the molecule and it is shown that the distorted ring shape in this structure is one which gives good matching of these orbitals. The P–OMe bonds (average 1♵6 Å) are also shorter than expected for a single bond. The shortening of this value from the accepted P–N single bond length (1♷7 Å) indicates some π bonding between phosphorus and nitrogen arising from donation of the nitrogen lone-pair electrons to vacant 3 d orbitals on phosphorus.
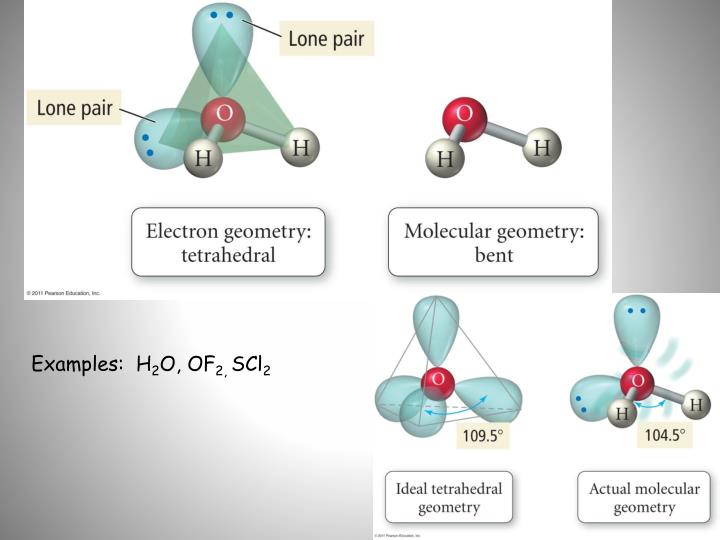
For instance, in BeCl2 there are only two electron pairs around Be, so the farthest they can get from each other is to be on opposite sides of Be, so the angle between them will be 180 and the geometry of the molecule will be linear. The six-membered ring of alternate phosphorus and nitrogen atoms has a twisted boat shape with an average P–N bond length of 1♶6 Å. The resulting geometry is determined by whatever arrangement gives the lowest repulsion energy. The atomic positions have been determined from X-ray diffraction data. Crystals of 2,4,6-trimethoxy-1,3,5-trimethyl-2,4,6-trioxocyclotriphosphazane, N 3Me 3P 3O 3(OMe) 3, are monoclinic, a= 11♳2, b= 8♲9, c= 7♶3 Å, β= 98♳°, space-group P2 1 with two molecules in the unit cell.
